Effect of Temperature
In leak testing using pressure-measuring methods, the temperature of the air test medium has a significant influence on the measurement result. This is essentially influenced by the temperature of the test specimen and the ambient temperature, in addition to the temperature of the air preparation.
The test procedure usually consists of 4 phases: Filling, Stabilization, Testing, and Emptying. The temperature change during the test phase, i.e., the period in which the pressure change is measured and evaluated, is crucial.
If the temperature of the air in the test specimen rises during the test phase, e.g., due to a warm test specimen, the pressure increases. A pressure reduction caused by a leak can, under certain circumstances, be compensated for, which could lead to a leaky part being evaluated as “tight.”
If the temperature of the air in the test specimen drops during the test phase, e.g., due to cold drafts, the pressure decreases. A pressure reduction due to a leak can thus be so greatly amplified that an actually tight part is evaluated as “leaky.”
The extent of temperature’s influence on the measurement result will be illustrated using the following test parameters as an example.
|
Test Pressure: |
p = 2000 mbar abs. (corresponds to 1000 mbar gauge pressure) |
|
Test Specimen Volume: |
V = 110 cm³ |
|
Permissible Leak Rate: |
Q = 0.5 cm³/min |
|
Test Time: |
5 sec |
|
Test Air Temperature: |
t = 23°C |
The pressure change ∆pL due to a leak is approximately calculated using the following formula:

In our example:
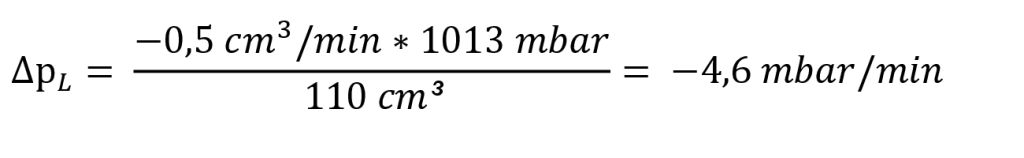
for a test time of 5 sec:

The pressure change ∆pT due to a temperature change is approximately calculated using the following formula:
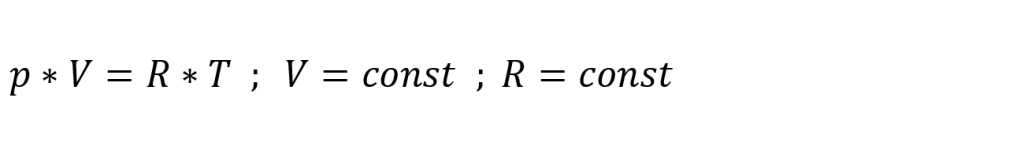
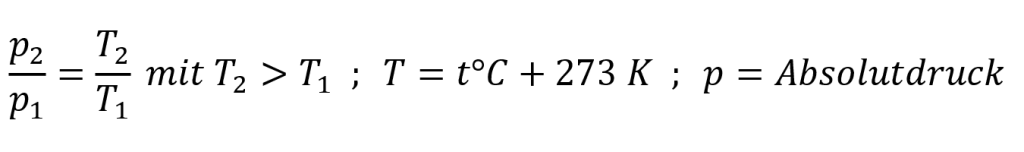
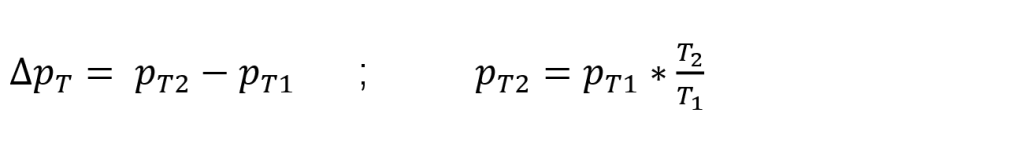

Accordingly, in our example, a temperature increase of 0.1°C during the 5 sec test time yields the following calculation:
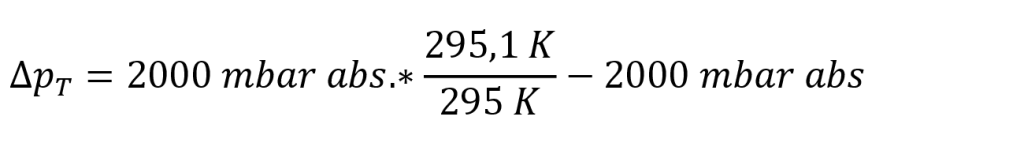

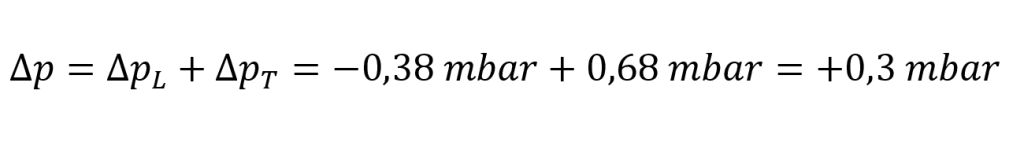
Result:
In our example, the test pressure would increase by 0.3 mbar, even though a leak of 0.5 cm³/min is present.
This effect is usually counteracted by a preceding stabilization phase. The stabilization time must be chosen such that temperature equalization between the test specimen and the test air is achieved. In this way, a temperature change during the subsequent test phase can be kept very low. Furthermore, the test time itself can be extended to achieve a larger pressure drop.